One of our previous blogposts discussed medical device industry trends and innovations.
Since then, more recent data on the number of patent applications from the European Patent Organisation’s (EPO) yearly research were made available. These can be used as a measure for innovation within the medical technology sector and other sectors.
Contrary to the expectations of many, the medical technology industry is still leading the pack, beating digital communication and computer technology. According to this research, 14.295 med tech patent applications were filed in 2020 [1].
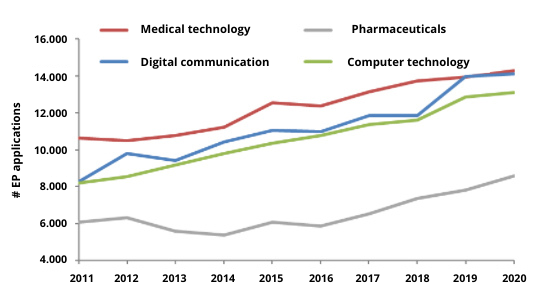
Figure 1 – # EP applications based on EPO data [1]
Increasingly, software is taking center stage, both in and around medical devices. The acronyms SaMD (Software as a Medical Device), SiMD (Software in a Medical Device) and MDSW (Medical Device Software) are becoming increasingly familiar.
Nowadays, devices are “connected” thanks to the rise of Internet of Things (IoT) and its components (smartphones, wearables, cloud-servers, cheaper and better sensor technology, big data, and AI). The Covid pandemic further accelerated this medical device industry trend and necessitated remote healthcare services.
In this blog post, we dive a little deeper into some of these medical device industry trends and technologies and their application to the medical industry.
4 major medical device industry trends
Sensors and Wearables
Thanks to the miniaturization and cost-effectiveness of sensor technology, sensors are built into all kinds of body-worn formats, like patches, watches, or bandages. These wearables are promoting increasing use in the continuous monitoring of health conditions.
For example, these sensors can monitor a patient’s clinical condition and these data, in turn, can even be used by software to determine the need for an intervention.
Please note that not all these wearable devices can be classified as Medical Devices. A lot depends on their intended use. Consider your smartwatch that monitors your heart rate for fitness-related purposes or tracks your sleep pattern.
For these types of uses, the manufacturer typically claims that the results cannot be used for any clinical purpose. Thus, he does not need to comply with medical device regulations. In case the sensor data are used for clinical decision-making, these devices must comply with applicable medical device regulations.
Artificial Intelligence (AI)
One of the most profound changes in healthcare today is the use of medical devices that incorporates software with Artificial Intelligence (AI) and Machine Learning (ML).
Think about: glucometers in controlling diabetes, and their possibility of optimizing insulin delivery, or software that is trained to detect arrhythmia in electrocardiograms or image-based diagnostics.
However, incorporating these technologies in medical devices is also one of the most profound challenges, not only from a technical standpoint but certainly from a regulatory standpoint as well.
This is also recognized by the FDA, which recently issued a regulatory action plan.
Guidance on the application of European medical devices regulations to devices incorporating these technologies is still missing.
Therefore, we’ve created a separate blog post that dives deeper into this topic.
Diagnostics and Point-of-Care testing
In the age of coronavirus, medical devices have captured the public imagination like never before. Ventilators, respirators, and face masks have probably saved thousands of lives. In addition, the virus also accelerated the medical device trend to bring diagnostics closer to the patient, so-called Point-of-Care (POC) testing.
Unlike traditional diagnostics, where samples from the POC are sent to centralized laboratories and people have to wait for hours/days/weeks to get the results, POC tests are performed at a physician’s office, at the pharmacy or even at home.
POC testing reduces the burden of sample logistics and allows us to focus on what matters, i.e., enabling rapid triage and clinical treatment decisions.
In doing so, POC testing systems benefit from the technologies described above. These devices can be connected to phones and can upload data to the cloud for physicians to review or to software for tracking trends and infectious disease outbreaks.
The Internet of (Medical) Things
All the technologies described in the sections above result in a huge amount of data. Devices are being connected and even legacy devices are being upgraded with connectivity capabilities.
All these data are collected and can be analysed, in real time if needed, to provide feedback to the patient, healthcare provider or can even be passed on to other connected medical devices for further analysis or as input.
It is believed that this connectivity, combined with the trend toward remote healthcare services, will play a crucial role in the future of healthcare.
Some experts envision what they call an Internet of Medical Things (IoMT) and describe it as “a connected infrastructure of medical devices, software applications, health systems, and services to a centralized healthcare IT system for more refined and in-depth data analysis” [2].
Nowadays data, and obviously also medical data, are highly valued. The confidentiality and privacy of such data require an entirely different discussion, but medical device companies, not to mention consumer technology companies like Google, Apple, and Amazon, are investing to make this happen:
- Apple’s CEO Tim Cook once said that “if you zoom out into the future, and you look back, and you ask the question ‘What was Apple’s greatest contribution to mankind?’ It will be about health.”
- Tech giant Samsung recently filed a patent with the EPO titled ‘Method and Electronic device for artificial intelligence (AI)-based assistive health sensing in the Internet of Things network’.
The overall IoMT concept and its exact interpretation may still seem somewhat vague and futuristic, but these medical device industry trends might just become reality sooner than you think.
Medical Device Industry Trends: end note
It is undeniable that the future of medical devices and med-tech, in general, looks exciting. However, medical device companies must divide their attention among a broad variety of tasks. Not only do the technical and innovative aspects of their product require their attention, but also the regulatory and quality aspects are challenging and, in some cases, underestimated.
Under the new EU Medical Device regulations, i.e., regulations 2017/745 (MDR) that is applicable since 26 May 2021 and regulation 2017/746 (IVDR) that will be applicable as of 16 May 2022, risk classification and defining the regulatory requirements for devices containing software and new technologies is not always straightforward.
In addition, medical device manufacturers are expected to follow industry standards and guidelines applicable for software (e.g., ISO13485, ISO 14971, IEC 62304, IEC 82304 …) not to mention, cybersecurity.
At QbD, we pride ourselves on tackling these kinds of challenges. We can help you determine the regulatory requirements and your strategy to align with them. So if you want more information, we are here for you. Don’t hesitate to get in touch with us.